In the previous post, “Frieda”, my coaching client, asked about producing test cases for auditors or regulators. In Rapid Software Testing (RST), we find it helpful to frame that in terms of formal testing.
Testing is formal to the degree that it must be done in a specific way, or to verify specific facts. Formal testing typically has the goal of confirming or demonstrating something in particular about the product. There’s a continuum to testing formality in RST. My version, a tiny bit different from James Bach‘s, looks like this:
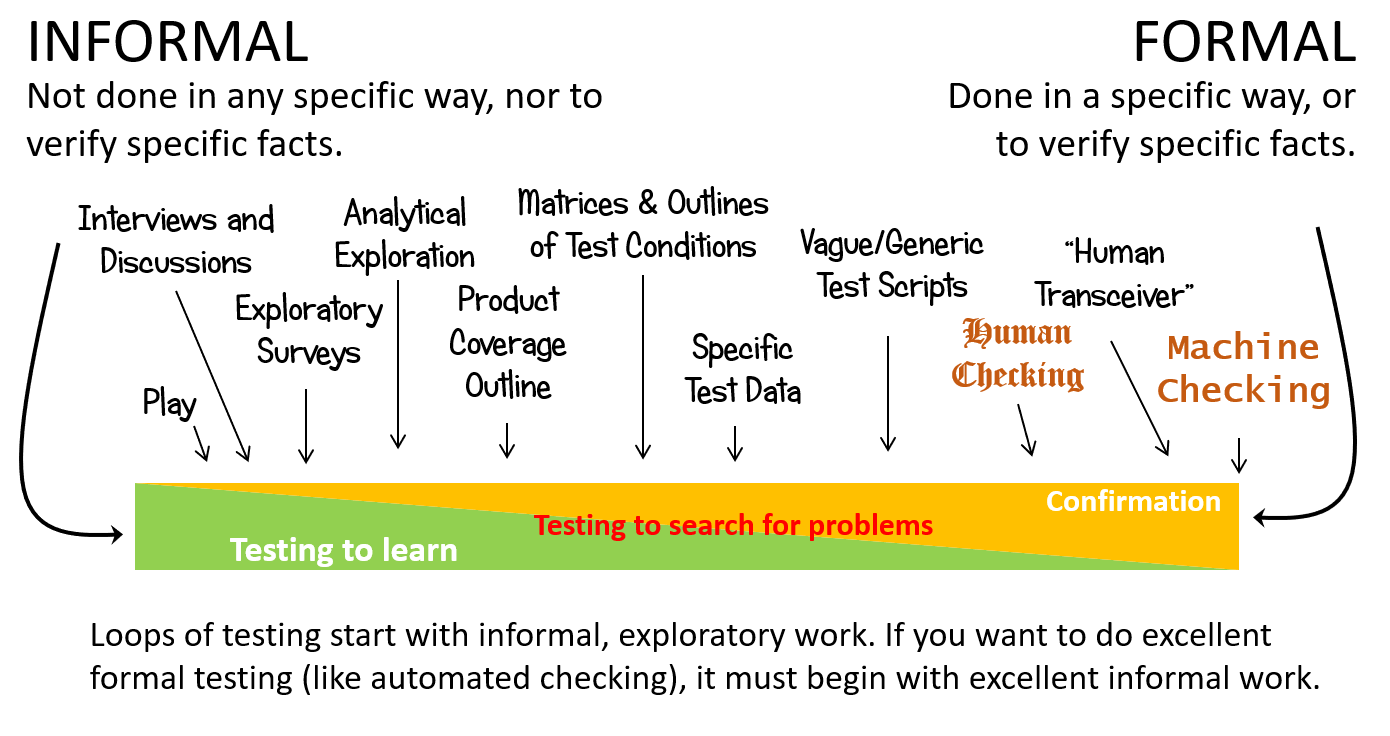
Some terminology notes:
- Checking is the process of operating and observing a product; applying decision rules to those observations; and then reporting on the outcome of those rules; all mechanistically, algorithmically. A check can be turned into a formally scripted process that can be performed by a human or by a machine.
- Procedurally scripted test cases are instances of human checking, where the tester is being substantially guided by what the script tells her to do. Since people are not machines and don’t stick to the algorithms, people are not checking in the strictest sense of our parlance.
- A human transceiver is someone doing things based only on the instructions of some other person, behaving as that person’s eyes, ears, and hands.
- Machine checking is the most formal mode of testing, in that machines perform checks in entirely specific ways, according to a program, entirely focused on specific facts. The motivation to check doesn’t come from the machine, but from some person.
Notice that programs are formal, but programming is an informal activity. Toolsmiths and people who develop automated checks are not following scripts themselves.
The degree to which you formalize is a choice, based on a number of context factors. Your context guides your choices, and both of those evolve over time.
One of the most important context factors is your mission. You might be in a regulated environment, where regulators and auditors will eventually want you to demonstrate specific things about the product and the project in a highly formal way. If you are in that context, keeping the the auditors and the regulators happy may require certain kinds of formal testing. Nonetheless, even in that context, you must perform informal testing—lots of it—for at least two big reasons.
The first big reason is to learn the about the product and its context to prepare for excellent formal testing that will stand up to the regulators’ scrutiny. This is tied to another context factor: where you are in the life of the project and your understanding of the product.
Formal testing starts with informal work that is more exploratory and tacit, with the goal of learning; less scripted and explicit, with the goal of demonstrating. All the way along, but especially in between those poles, we’re searching for problems. No less than the Food and Drug Administration emphasizes how important this is.
Thorough and complete evaluation of the device during the exploratory stage results in a better understanding of the device and how it is expected to perform. This understanding can help to confirm that the intended use of the device will be aligned with sponsor expectations. It also can help with the selection of an appropriate pivotal study design.
Section 5: The Importance of Exploratory Studies in Pivotal Study Design;
Design Considerations for Pivotal Clinical Investigations for Medical Devices
Guidance for Industry
Clinical Investigators, Institutional Review Boards and Food and Drug Administration Staff
The pivotal stage of device development, says the FDA, focuses on developing what people need to know to evaluate the safety and effectiveness of a product. The pivotal stage usually consists of one or more pivotal studies. In other words, the FDA acknowledges that development happens in loops and cycles; that development is an iterative process.
James Bach emphasized this in his talk The Dirty Secret of Formal Testing and it’s an important point in RST. Development is an iterative process because at the beginning of any cycle of work, we don’t know for sure what all the requirements are; what they mean; what we can get; and how we might decide that we’ve got it. We don’t really know that until we’ve until we’ve tested the product… and we don’t know how to test the product until we’ve tried to test the product!
Just like developing automated checks, developing formally scripted test cases is an informal process. You don’t follow a script when you’re interpreting a specification; when you’re having a conversation with a developer or a designer; when you’re exploring the product and the test space to figure out where checking might be useful or important. You don’t follow a script when you recognize a new way of using tools to learn something about the product, and apply them. And you don’t follow a script when you investigate bugs that you’ve found—either during informal testing or the formal testing that might follow it.
If you try to develop formal procedural test cases without testing the actual product, they stand a good chance of being out of sync with it. The dirty secret of formal testing is that all good formal testing begins with informal testing.
It might be a very good idea for programmers to develop some automated checks that helps them with the discipline of building clean code and getting rapid feedback on it. It’s also a good idea for developers, designers, testers, and business people to develop clear ideas about intentions for a product, envisioning success. It might also be a good idea to develop some automated checks above the unit level and apply them to the build process—but not too many and certainly not too early. The beginning of the work is usually a terrible time for excessive formalization.
Which brings us to the second big reason to perform informal testing continuously throughout any project: to address the risk that our formal testing to date will fail to reveal how the product might disappoint customers; lose someone’s money; blow something up; or hurt or kill people. We must be open to discovery, and to performing the testing and investigation that supports it, all the way throughout the project, because neither epiphanies nor bugs follow scripts or schedules.
The overarching mission of testing is focused on a question: “are there problems that threaten the value of the product, or the on-time, successful completion of our work?” That’s not a question that formal testing can ever answer on its own. Fixation on automated checks or test cases runs the risk of displacing time for experimentation, exploration, discovery, and learning.
Next time, we’ll look at an example of breaking test case addiction on a real medical device project. Stay tuned.
[…] Breaking the Test Case Addiction (Part 3) Written by: Michael Bolton […]